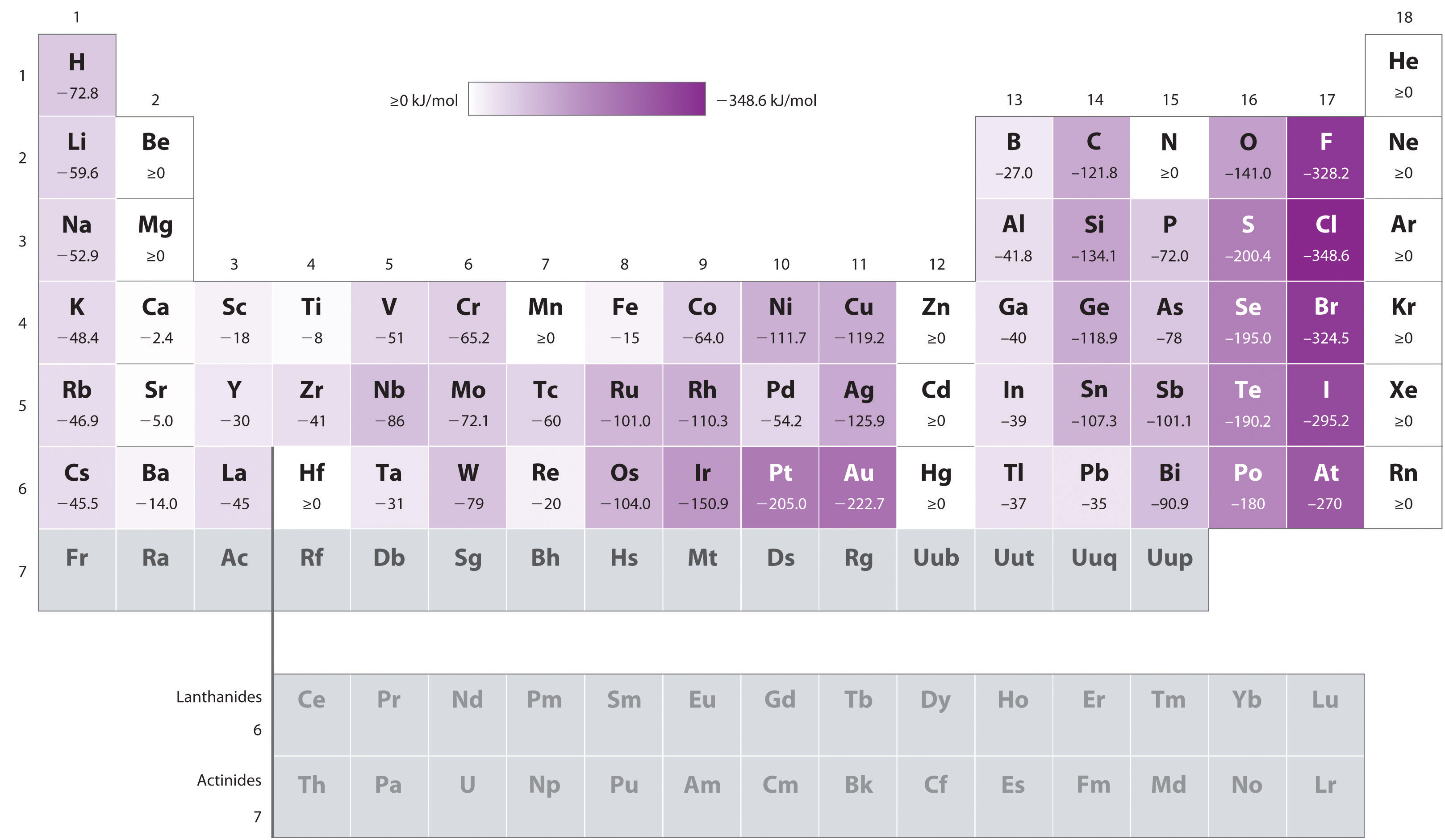
This reduces the electron-electron repulsions, making it more attractive for an electron to be gained. In the chloride anion, however, the new electron is added into the third shell, occupying more space. But why?įluorine is the smallest atom of the halogens and an incoming electron experiences a significant repulsion from the electrons already present. In halogens, chlorine has a more negative electron affinity value than fluorine. Incoming electrons, therefore, experience less nuclear attraction leading to less negative electron affinities. Moving down group 1, the atomic size increases as the electrons occupy higher principal quantum numbers. Electron affinities, similar to ionization energies, show trends in the periodic table. In general, the greater the attraction between an atom and an added electron, the more negative the electron affinity. 
The negative sign indicates that it is an exothermic change.Īrgon, however, has a positive electron affinity, indicating that formation of an argon anion requires energy to be supplied. Electron affinity measures the ease of gaining an electron by an atom.įor example, the electron affinity of chlorine is −348.6 kJ/mol. When an electron is added to a gaseous atom, a change in energy is observed called electron affinity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
